Mitofusin's fusion-independent roles in proteostasis surveillance
Researchers have discovered a novel connection between mitochondrial function, protein quality control and cellular health, whose failure could be the leading cause in the currently incurable neurological disease Charcot-Marie-Tooth (CMT).
The study uncovered an unexpected function of the protein Mitofusin 2 (MFN2) in mitochondria. It could have far-reaching implications for the treatment of CMT and similar conditions. The work was published in Nature Communications under the title ‘Mitofusin 2 displays fusion-independent roles in proteostasis surveillance’.
Mitochondria are best known as the energy producers of the cell, but they also regulate metabolism, gene expression and cell survival, all essential for healthy aging. MFN2 has long been recognized for its role in mitochondrial fusion.
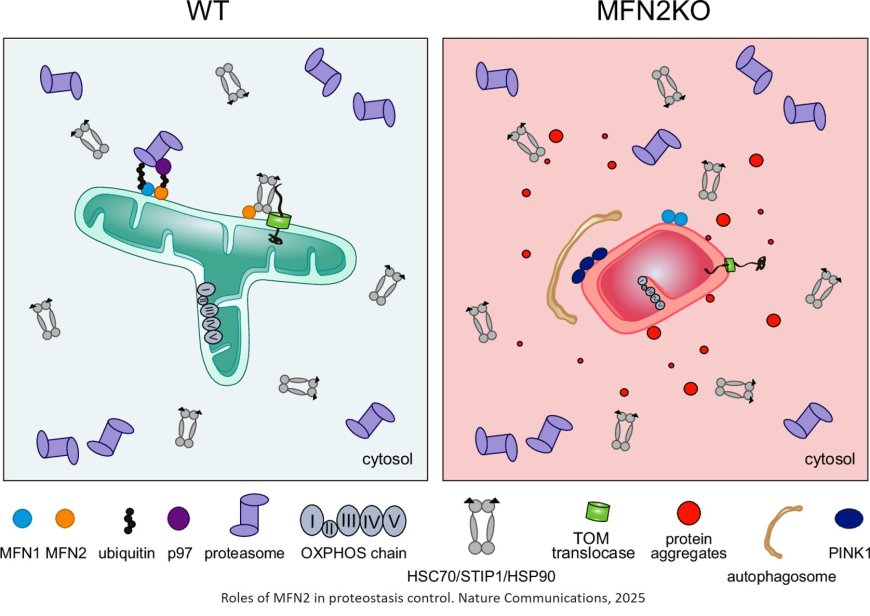
The researchers now uncovered an additional, unexpected function of MFN2 in maintaining protein quality within cells. The team found that MFN2 interacts with the proteasome and chaperones – cellular systems that prevent newly produced proteins from forming toxic aggregates, which can lead to neurodegeneration. Unexpectedly, MFN2 shares this protective function. Further analysis of skin cells from CMT patients confirmed that when MFN2 is mutated, this novel function is lost, leading to harmful protein clumping. This discovery opens new possibilities for treating diseases like CMT.
“Although MFN2 is a leading causative gene in Charcot-Marie-Tooth, most other genes linked to the disease do not encode mitochondrial proteins. This makes it less surprising that MFN2’s connection to CMT is independent of its primary function in mitochondrial dynamics,” explained one of the first authors.
To understand MFN2’s unique role, the researchers compared it to its closely related counterpart, MFN1. While hundreds of mutations in MFN2 are known to cause CMT, MFN1 has not been linked to the disease. By generating human cell lines lacking either MFN1 or MFN2, they found that only MFN2 interacts with the proteasome and prevents harmful protein accumulation.
“The study suggests that MFN2’s function in protein quality control could also be relevant for diseases like obesity, where cellular stress and protein misfolding play a major role,” said another author on the study.
According to the researchers, this discovery is a key step in uncovering how mitochondria contribute to cellular health. The team now sees potential for new therapeutic approaches.