Targeting GTPase family of proteins!
Scientists have discovered how to target a class of molecular switches called GTPases that are involved in a myriad of diseases from Parkinson’s to cancer and have long been thought to be “undruggable.”
Because of their slippery exteriors, the GTPases have remained largely out of reach of modern drug discovery, with the exception of the notorious cancer-causing GTPase called K-Ras.
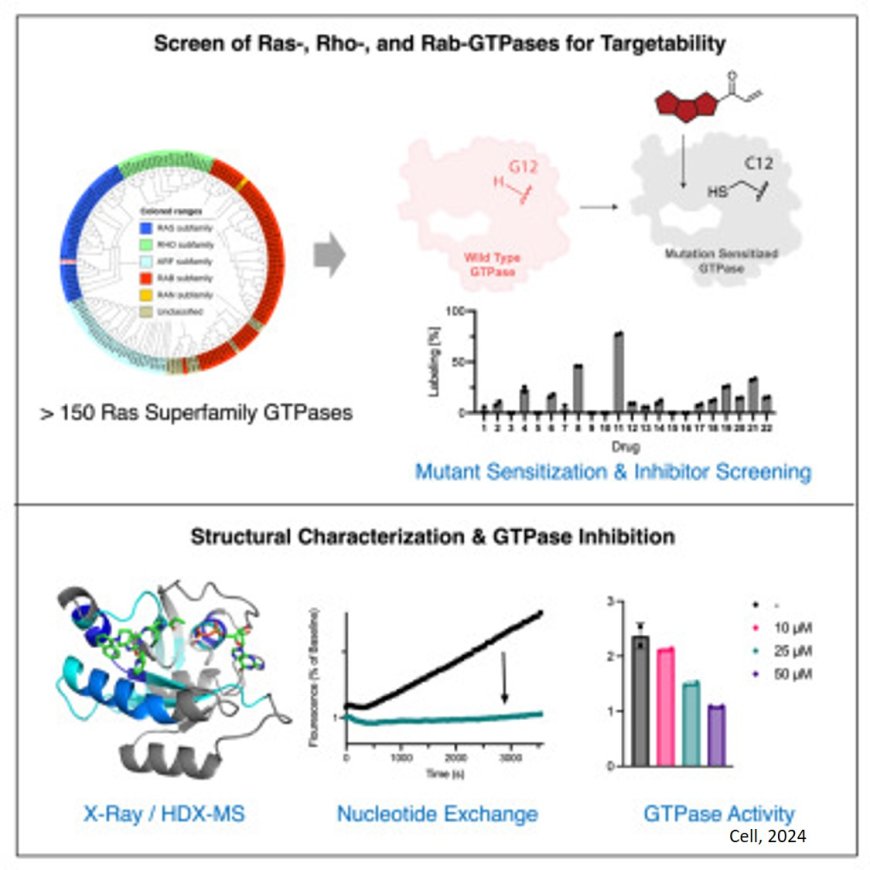
On a hunch, the team tested a dozen drugs that target K-Ras against a handful of GTPases they had mutated to make them more receptive to the drugs. The approach revealed new drug binding sites that could not have been predicted by computational drug discovery tools.
The findings, published in Cell, create a long-awaited opportunity to develop therapies for the many, diverse diseases that arise from GTPase dysfunction.
“We’ve known about the GTPases for decades but have lacked any way to reliably drug them,” said the senior author of the paper. “This really puts all those GTPases on the map for drug discovery, so it’s possible to target them when they’re associated with disease.”
Our cells depend on networks of GTPases, which oversee everything from the movement of molecules to cell growth and division. When something goes wrong among these switches, disease can follow.
In 2013, the researchers discovered a “pocket” where drugs could bind to K-Ras, a famous GTPase that’s responsible for up to 30% of all cancer cases.
Since then, nearly a dozen drugs have been developed targeting mutations to K-Ras, but the other GTPases remained untouchable.
In the present work, the team engineered one of the cancer-causing K-Ras mutations, G12C, into a representative group of GTPases.
They guessed that G12C, which places a chemical “hook” onto a protein, could help them fish out which among ten K-Ras G12C drugs might bind to other GTPases, which have only subtle similarity to K-Ras itself.
The laboratory experiments turned up gold: with the help of G12C, some of the K-Ras drugs bound to the otherwise featureless GTPases. When G12C was removed, those drugs still bound to the GTPase.
The approach, dubbed chemical genetics, exploited the flexibility of the GTPases, enabling the drugs to nudge open a pocket in the protein where it could lodge itself. This pocket had evaded previous efforts to predict, computationally, where drugs might bind.
“Since these GTPases switch between ‘on’ and ‘off’ states, the pocket is not usually visible, certainly not to the standard software used for drug discovery,” the author said. “Instead, the drug binds to an intermediate state, freezing the GTPases and inactivating them.”
The researchers are sharing their methods openly in the hopes that others will use them to drug their GTPase of interest, whether it’s a Rab GTPase, which is implicated in Alzheimer’s, or a Rac GTPase, which plays a role in breast cancer. Among the hundreds of GTPases, there’s rich potential to make progress for patients.
“In the case of these enzymes, it was critical for us to first test our ideas experimentally in the laboratory, to actually see what worked,” the author said. “We’re hopeful it can really accelerate drug discovery.”