Noninvasive monitoring of gene expression in primate brains
Gene therapy has been successfully used to treat a number of diseases, including immune deficiencies, hereditary blindness, hemophilia and, recently, Huntington’s disease, a fatal neurological disorder.
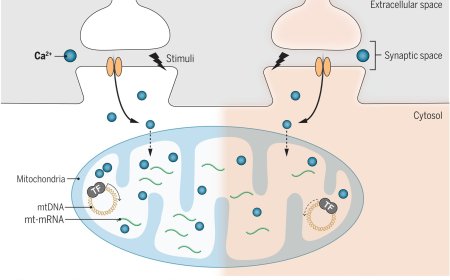
An advance reported in the journal Neuron adds to the technique’s growing track record of evidence supporting the view that it could unlock powerful, personalized therapies: researchers found that released markers of activity (RMAs) ⎯ engineered proteins designed to cross the blood-brain barrier and persist in the blood for hours at a time, providing a reliable and noninvasive way to get information about gene expression in the brain ⎯ work just as well in monkeys as they do in mice.
On the route from laboratory discovery to lifesaving treatment, large animal model studies are a critical part of the process. Most research never reaches this stage.
“Our study shows it is fairly easy to translate this noninvasive technique between species,” an author said. “This is exciting because RMAs are an extremely sensitive tool that could be used to track as few as tens to hundreds of neurons at a time ⎯ no existing imaging or monitoring technique can give us that level of precision.”
Alongside precision, RMA technology is also capacious and adaptable: Different serum markers can be designed to track multiple genes across different brain regions.
“Protein detection can be multiplexed,” the author said. “In the future, it should be possible to detect a large number of different synthetic serum markers in a single sample using a variety of biochemical techniques, such as mass-spectrometry or single-molecule protein sequencing.”
Monitoring gene expression in the living, intact brain could reveal critical information about cellular activity, complex cognitive processes and how neurological disease starts and progresses. By retrieving this information using a simple blood test, tracking the same individual brain over time becomes feasible.
“In brain research, longitudinal monitoring is especially important,” the author said, giving addiction as an example. “Terminal or biopsy readouts are snapshots. By monitoring the same individual over time we can see the downstream effects of gene expression and how they shape future disease or physiology.
“To understand conditions like addiction, you need more than a single snapshot of the brain. We need to see the movie, not just a photograph. Tracking the living brain over time lets us actually watch which genes drive these changes as they happen.”
The author developed the RMA platform based on the observation that antibody therapies failed because antibodies were quick to migrate from the brain into the blood. The author zeroed in on the part of antibodies that allows them to cross the blood-brain barrier and used it as a building block for the synthetic reporters.
“It is a short piece of a protein that is responsible for the exit of the protein from the cell into the space between cells, into the extracellular matrix,” the author said. “Simply changing the mouse version of this protein domain for the rhesus macaque version was enough to make the reporter functional in the other species.”