Human-specific genes regulate key gene mutated in autism spectrum disorders
The human brain’s remarkably prolonged development is unique among mammals and is thought to contribute to our advanced learning abilities. Disruptions in this process may explain certain neurodevelopmental diseases. Now, a team of researchers has discovered a link between two genes, present only in human DNA, and a key gene called SYNGAP1, which is mutated in intellectual disability and autism spectrum disorders.
Their study, published in Neuron, provides a surprisingly direct link between human brain evolution and neurodevelopmental disorders.
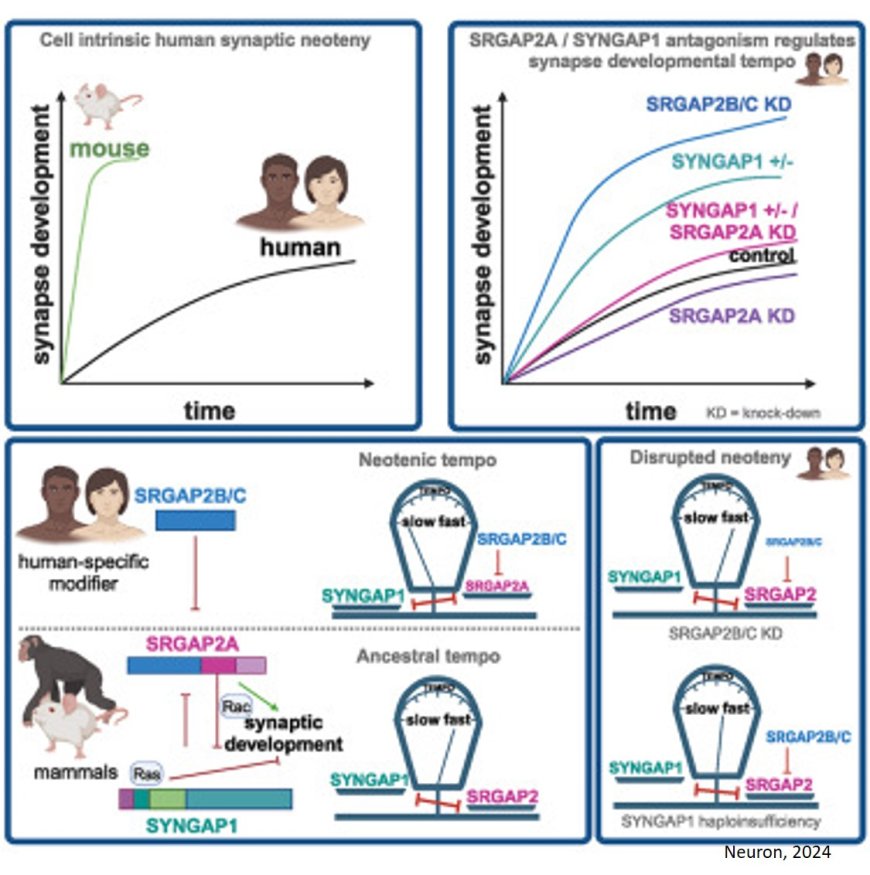
The human brain stands out among mammals for its remarkably prolonged development. Synapses – critical connections between neurons of the cerebral cortex, the brain’s main hub for cognition – take years to mature in humans, compared to just months in species like macaques or mice. This extended development, also known as neoteny, is thought to be central to humans' advanced cognitive and learning abilities. On the other hand, it has been hypothesized that disruptions of brain neoteny could be linked to neurodevelopmental disorders such as intellectual disability and autism spectrum disorder.
The lab previously discovered that the prolonged development of the human cerebral cortex is mainly due to human-specific molecular mechanisms in neurons. Now, they are investigating these molecular timers in human neurons.
In their latest study, the team tested the involvement of two genes, SRGAP2B and SRGAP2C, which are unique to humans. These genes have been found to slow down synapse development when artificially introduced into mouse neurons of the cerebral cortex. The question if these genes function the same way in human neurons has remained unanswered.
The researchers switched off SRGA2B and SRGAP2C in human neurons, transplanted them into mouse brains, and carefully monitored synapse development over an 18-month period.
“We discovered that when you turn off these genes in human neurons, synaptic development speeds up at remarkable levels," says the author. "By 18 months, the synapses are comparable to what we would expect to see in children between five and ten years old! This mirrors the accelerated synapse development observed in certain forms of autism spectrum disorder.”
The team then investigated the underlying genetic mechanisms behind the pronounced effects of SRGAP2B and SRGAP2C on human neuron neoteny. They focused on the SYNGAP1 gene, an important disease gene known to be involved in intellectual disability and autism spectrum disorder.
Remarkably, they discovered that the SRGAP2 and SYNGAP1 genes act together to control the speed of human synapse development. Most strikingly, they found that SRGAP2B and SRGAP2C increase the levels of the SYNGAP1 gene and can even reverse some defects in neurons lacking SYNGAP1. This finding increases our understanding of how human-specific molecules influence neurodevelopmental disease pathways, shedding light on why such disorders are more prevalent in our species.