Genomic regions with epigenetic modifications linked to ALS progression identified!
For most patients, it’s unknown exactly what causes amyotrophic lateral sclerosis (ALS), a disease characterized by degeneration of motor neurons that impairs muscle control and eventually leads to death.
Studies have identified certain genes that confer a higher risk of the disease, but scientists believe there are many more genetic risk factors that have yet to be discovered. One reason why these drivers have been hard to find is that some are found in very few patients, making it hard to pick them out without a very large sample of patients. Additionally, some of the risk may be driven by epigenomic factors, rather than mutations in protein-coding genes.
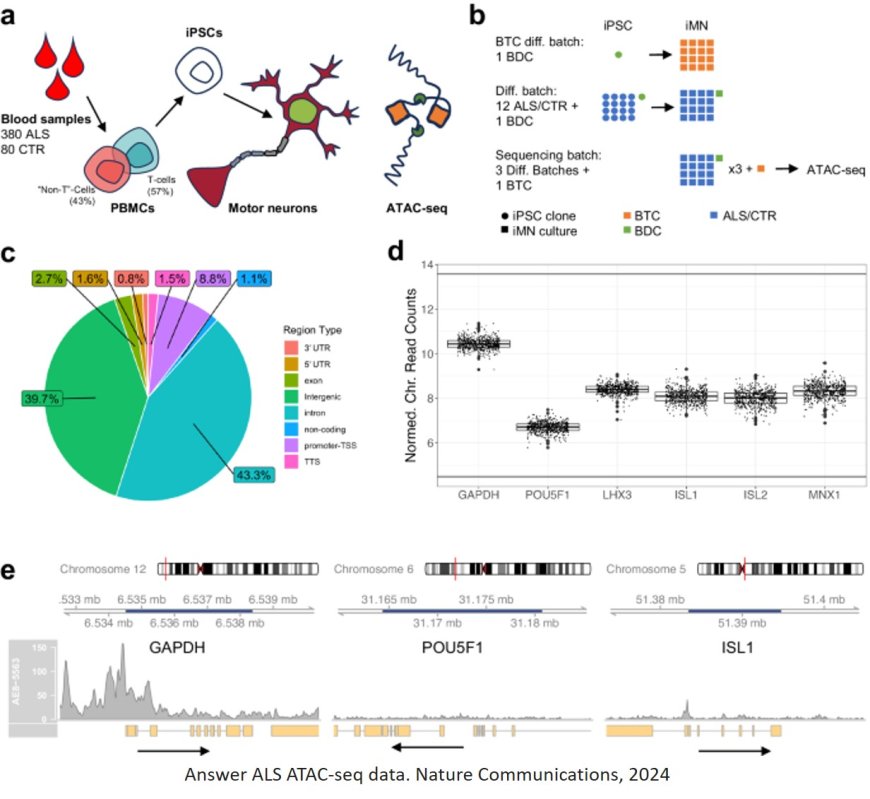
Working with the Answer ALS consortium, a team of researchers has analyzed epigenetic modifications — tags that determine which genes are turned on in a cell — in motor neurons derived from induced pluripotent stem (IPS) cells from 380 ALS patients.
This analysis revealed a strong differential signal associated with a known subtype of ALS, and about 30 locations with modifications that appear to be linked to rates of disease progression in ALS patients. The findings may help scientists develop new treatments that are targeted to patients with certain genetic risk factors.
“If the root causes are different for all these different versions of the disease, the drugs will be very different and the signals in IPS cells will be very different,” says the senior author of the study. “We may get to a point in a decade or so where we don’t even think of ALS as one disease, where there are drugs that are treating specific types of ALS that only work for one group of patients and not for another.”
ALS is a rare disease that is estimated to affect about 30,000 people in the United States. One of the challenges in studying the disease is that while genetic variants are believed to account for about 50 percent of ALS risk (with environmental factors making up the rest), most of the variants that contribute to that risk have not been identified.
Similar to Alzheimer’s disease, there may be a large number of genetic variants that can confer risk, but each individual patient may carry only a small number of those. This makes it difficult to identify the risk factors unless scientists have a very large population of patients to analyze.
“Because we expect the disease to be heterogeneous, you need to have large numbers of patients before you can pick up on signals like this. To really be able to classify the subtypes of disease, we’re going to need to look at a lot of people,” the author says.
About 10 years ago, the Answer ALS consortium began to collect large numbers of patient samples, which could allow for larger-scale studies that might reveal some of the genetic drivers of the disease. From blood samples, researchers can create induced pluripotent stem cells and then induce them to differentiate into motor neurons, the cells most affected by ALS.
“We don’t think all ALS patients are going to be the same, just like all cancers are not the same. And the goal is being able to find drivers of the disease that could be therapeutic targets,” the author says.
In this study, the researchers wanted to see if patient-derived cells could offer any information about molecular differences that are relevant to ALS. They focused on epigenomic modifications, using a method called ATAC-seq to measure chromatin density across the genome of each cell. Chromatin is a complex of DNA and proteins that determines which genes are accessible to be transcribed by the cell, depending on how densely packed the chromatin is.
In data that were collected and analyzed over several years, the researchers did not find any global signal that clearly differentiated the 380 ALS patients in their study from 80 healthy control subjects. However, they did find a strong differential signal associated with a subtype of ALS, characterized by a genetic mutation in the C9orf72 gene.
Additionally, they identified about 30 regions that were associated with slower rates of disease progression in ALS patients. Many of these regions are located near genes related to the cellular inflammatory response; interestingly, several of the identified genes have also been implicated in other neurodegenerative diseases, such as Parkinson’s disease.
“You can use a small number of these epigenomic regions and look at the intensity of the signal there, and predict how quickly someone’s disease will progress. That really validates the hypothesis that the epigenomics can be used as a filter to better understand the contribution of the person’s genome,” the author says.
The researchers now hope to further investigate these genomic regions and see how they might drive different aspects of ALS progression in different subsets of patients. This could help scientists develop drugs that might work in different groups of patients, and help them identify which patients should be chosen for clinical