Obesity leaves a lasting memory in immune cells
People who live with obesity are ‘tagging’ a memory of being overweight on a key part of the immune system - leaving people with ongoing risk of obesity-related conditions years after losing weight, according to a 10 year-long study published in EMBO Reports.
A European research team have shown that immune cells known as helper T cells (or CD4+ lymphocytes) carry a long memory of obesity.
Through a process known as DNA methylation, tags or markers attach themselves to DNA in the immune cells. This ‘tagging’ is likely to last between 5-10 years after people successfully lose weight. The resulting impact of the ‘memory’ of obesity in helper T cells could cause dysregulation of some usual activities that the immune system typically does, including cleaning away waste and regulating immune ageing.
The research team believe that this could leave people who do lose weight to continue to be at risk of obesity-related conditions long after reaching a normal weight.
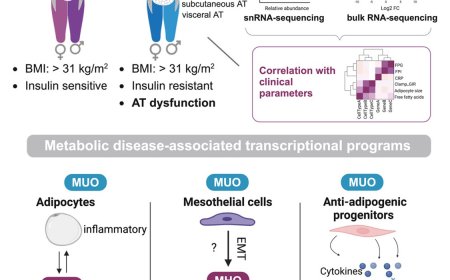
Researchers took immune cells from four groups of people to provide the most detailed picture of the impact of obesity. The study included:
· Collecting blood from patients who were living with obesity and received weight loss injections,
· Collecting blood from patients with a rare genetic disorder called Alstrom Syndrome characterised by early-onset childhood obesity, and healthy matched pairs,
· Collecting blood and fat tissue from a cohort of participants who took part in a 10-week exercise intervention
· Collecting blood and fat tissue from either normal weight or obesity cohorts with osteoarthritis undergoing total hip or knee replacement surgery.
The work additionally studied cells from mice models fed on a high-fat diet, and blood donations from healthy human volunteers. These models were used to help look at the mechanisms within cells that underpin immune dysregulation in obesity.
A co-lead author of the study said: “The findings suggest that short-term weight loss may not immediately reduce the risk of some disease conditions associated with obesity, including type 2 diabetes and some cancers.
“Instead, ongoing weight management following loss will see the ‘obesity memory’ slowly fade. This may take several years of sustained weight loss maintenance, likely 5-10 years, though this requires further study, to fully reverse the effects of obesity on T cells.
“Additionally, our study suggests potential therapeutic opportunities to expedite this process, such as repurposing drugs like SGLT2 inhibitors, which have shown promise in reducing inflammation and promoting immune-mediated clearance of senescent cells in obesity.”
The author team identify two specific pathways through which tagging affects helper T cells.
The memory of obesity is thought to affect autophagy (where cells eat bits of themselves to clean away the waste) and immune senescence (which is ageing of the immune system). The research team will use these findings to help look for targeted treatments that help restore typical immune system functioning that is being inhibited by the DNA tagging. The resulting potential treatments could then be used alongside existing weight loss therapies to reduce the risk of conditions including metabolic diseases and cancer that are exacerbated by obesity.
The senior author of the paper said: “Our findings show that obesity is associated with durable epigenetic modifications that influence immune cell behavior. This suggests that the immune system retains a molecular record of past metabolic exposures, which may have implications for long-term disease risk and recovery.”
https://link.springer.com/article/10.1038/s44319-026-00765-w
https://sciencemission.com/DNA-methylation-mediated-memory-of-obesity